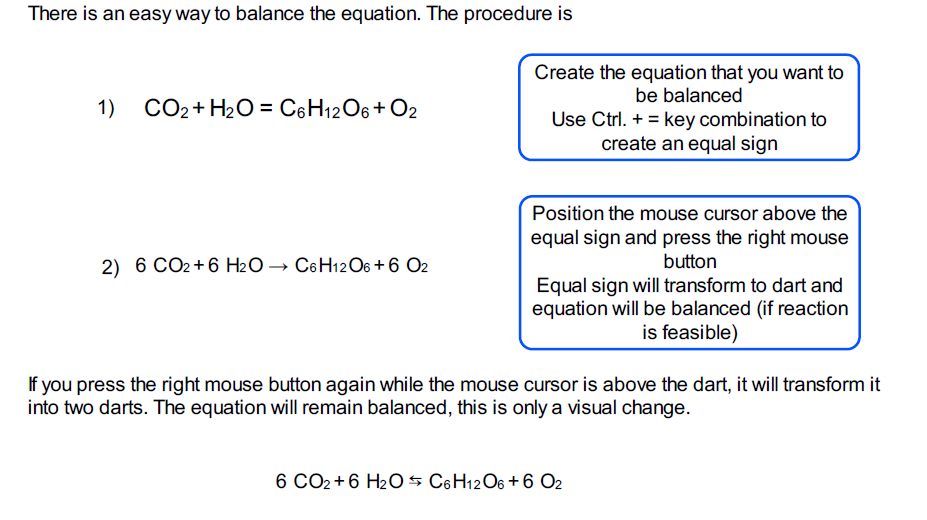
The solution to this problem is to put a 2 on the left. Count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges/ions) are balanced. As it is now, on the left is 1 Na, but on the right, there are 2 Na. Step 4: Substitute Coefficients and Verify Result. That's it! the reaction is now properly balanced □. To balance the above equation, which includes sodium and oxygen, this chemical equation must have the same amount of Na on the right side as well as on the left side. Mass of Reactants = Mass of Products \small \text+ 19\text O_2 \longrightarrow 12\text C\text O_2 + 14\text H_2\text O 2 C 6 H 14 + 19 O 2 ⟶ 12 C O 2 + 14 H 2 O Note: All reactants and products require a coefficient of at least one. Oxygen (O): 2 on the left, 3 on the right. Hydrogen (H): 4 on the left, 2 on the right. Make a tally for each element: Carbon (C): 1 on the left, 1 on the right. With this in mind, when balancing chemical reactions, the goal is to equilibrate the masses of reactants and products of the equation: Step 2: Count the number of atoms for each element: Examine each element in the equation and count the number of atoms on both sides. That means that the total mass of reactants and products is the same before and after the reaction. Enter an equation of a redox chemical reaction and press the Balance button. 
To balance the number of oxygen atoms, we can. For example, we can add a '2' coefficient in front of the water molecule on the product side to balance the number of hydrogen atoms: H2 + O2 -> 2H2O. 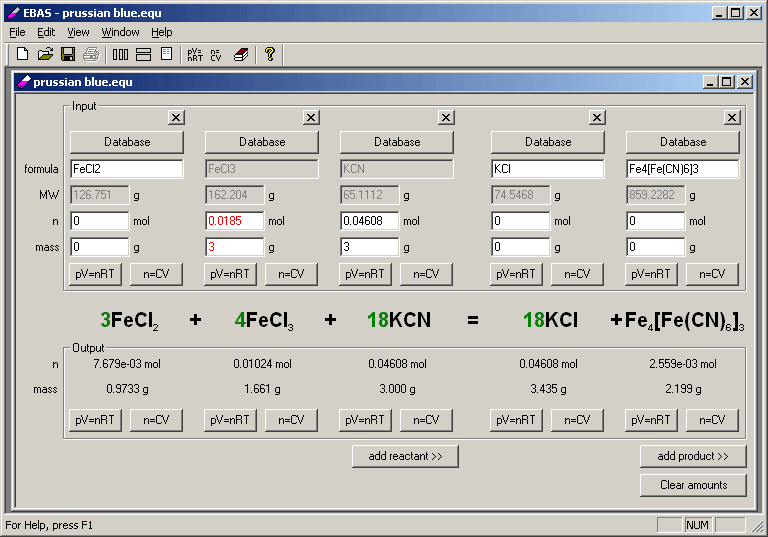
According to Lavoisier's law of conservation of mass, the total mass of a chemical reaction system doesn't change. This requires adding coefficients to each reactant and product in order to balance the number of atoms of each element on both sides of the equation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
